Cyanuric chloride
- 2,4,6-trichloro- 1 ,3,5-triazine
- Trichloro-s -triazine
- Cyanuric chloride
- Cyanuric
- Tricyanogenchlorid
- Trichlorcyanidin
- Cyanuryl chloride
White solid
Fixed
1.92 g · cm -3 ( 20 ° C)
146 ° C
194 ° C.
2.5 Pa ( 20 ° C)
Violent decomposition in water
Risk
Template: Infobox chemical / molecular formula search available
Cyanuric chloride is a reactive chemical, more specifically a heterocyclic compound. The eponymous basic building block is the 1,3,5- triazine (see also Hantzsch - Widman system) derived cyanuric acid (1,3,5- triazine-2 ,4,6 -triol ).
Production and representation
Cyanuric chloride is prepared in two steps by trimerization of cyanogen chloride. In the first step hydrocyanic acid is halogenated with chlorine to form cyanogen chloride, this then subsequently at elevated temperature trimerizes to a carbon contact:
2005, about 200,000 tons were produced worldwide.
Properties
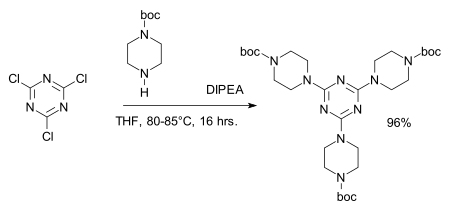
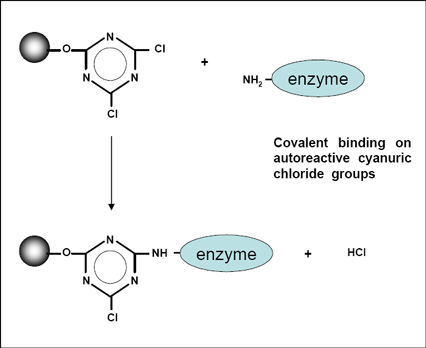
Cyanuric chloride is a white, fine crystalline powder. It irritates the mucous membranes and has a peculiar chlorine or chloride -like, pungent odor. In dry air at elevated temperature, it is stable; in the presence of traces of moisture is hydrolyzed to cyanuric acid and HCl. The rate of hydrolysis is highly dependent on temperature and pH. Into ice water buffered at pH = 7 as a suspension, it is stable for several hours, is carried out above 10 ° C with increasing temperature, a rapid increase in the hydrolysis, in which the hydrochloric acid formed during the hydrolysis acting autocatalytically. The three chlorine atoms in cyanuric chloride differ significantly in their reactivity towards nucleophiles. By appropriate choice of reaction conditions it is possible to selectively bring them with three different nucleophiles Nu1 to NU3 to the reaction:
Use
Cyanuric chloride is an important intermediate for the preparation of direct dyes, reactive dyes for cotton, optical brighteners and pesticides. The reactive dyes are derived from the dichlorotriazines and particularly the chlorotriazines.