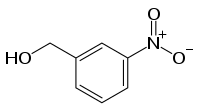
3-Nitrobenzyl alcohol
The Nitrobenzylalkohole form a group of substances derived from both the benzyl alcohol as well as from nitrobenzene in chemistry. The structure consists of a benzene ring with an added hydroxymethyl ( - CH2OH ) and nitro group (- NO2) as substituents. Due to their different arrangement (ortho, meta or para), three constitutional isomers with the formula C7H7NO3 result.
Representation
The Nitrobenzylalkohole can be shown by reduction with sodium borohydride ( NaBH4 ) of the nitrobenzoyl chlorides in anhydrous dioxane. They are also available by Meerwein Ponndorf - Verley reduction of the nitrobenzaldehydes. In basic medium the nitrobenzaldehydes disproportionate to nitrobenzoic acids and Nitrobenzylalkoholen.
Properties
The Nitrobenzylalkohole are solids. Of 4 -nitrobenzyl alcohol, which has the highest symmetry, has the highest melting point.
Use
In the mass spectrometry of 3 -nitrobenzyl alcohol is ( often referred to as "3- NBA " for short ) use as a liquid matrix for fast atom bombardment and matrix-assisted Laser-Desorption/Ionisation.