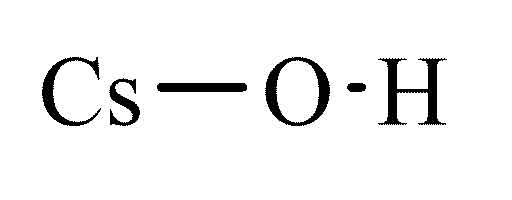Caesium hydroxide
- 21351-79-1 (anhydrous)
- 35103-79-8 (monohydrate)
White to yellowish crystals
Fixed
3.68 g · cm -3
272.3 ° C
Soluble in water
Risk
Template: Infobox chemical / molecular formula search is not possible
Cesium hydroxide, CsOH, is the hydroxide of cesium. It is one of the strongest bases in aqueous solution.
Production and representation
Cesium hydroxide formed in addition to hydrogen at the extremely violent reaction ( explosion) of water with cesium:
Another synthesis possibility is given by the reaction of cesium oxide with water.
During the mixing violent reactions may occur.
Properties
Aqueous solutions of cesium are basic solutions. Cesium and its aqueous solutions etch glass.
Use
The Caesiumlauge is a high quality base for the catalytic alkynylation of aldehydes and ketones.
Cesium hydroxide is used as an electrolyte in galvanic cells.